Consider This System At Equilibrium
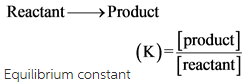
Consider this system at equilibrium. On this view a coherent theory of the price system and the coordination of economic activity has to consider the simultaneous general equilibrium of. If an object is at equilibrium then the forces are balanced. The equilibrium constant of a chemical reaction usually denoted by the symbol K provides insight into the relationship between the products and reactants when a chemical reaction reaches equilibrium.
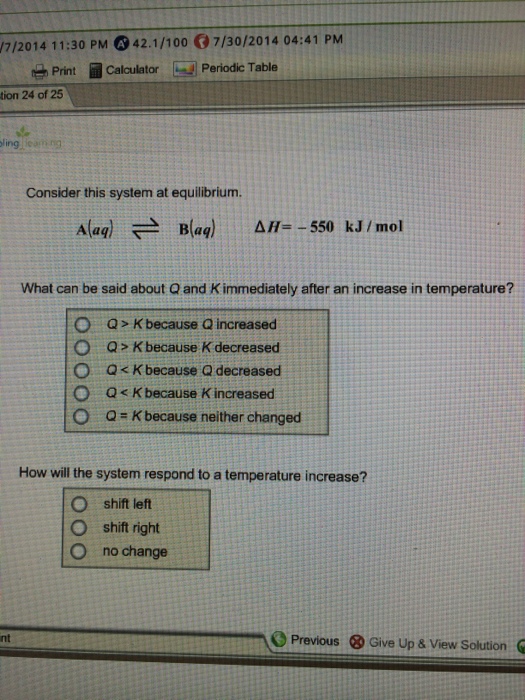
When they are thrown out of balance by a change in concentration temperature or pressure the system will naturally shift in such a way as to re-balance itself after the change. For example consider a flask with an equilibrium mixture of CO 2 CO and O 2 as in reaction 1. At iitill tii lPClA system initially containing only PCl 5 at t ti f 0 100t a concentration of 0100 M has a Q c 0 which is less than 0030.
Learn about Kc and Kp here. When physical forces are balanced in a system no further change occurs. Consider the two-dimensional autonomous system f1xy -y y-1 y f2xy x-1y-1 a Find all of the equilibrium points.
Thermodynamics is the study of the relationship between heat temperature work and energy. Vapor-liquid equilibrium VLE data can be computed using the modified Raoults law. Write out both the condition for zero net force and the condition for zero net torque.
Consider the following reaction CO2 g H2 g CO g H2O g Calculate the value of the equilibrium constant Kc for the above system if 01908 moles of CO2 00908 moles of H2 00092 moles of CO and 00092 moles of H2O vapour were present in a 200 L reaction vessel at equilibrium. CH 3 CO 2 H CH 3 CO 2 H Here in this case some protons will be released from acetic acid molecules during a forward reaction while the backward reaction tells us the formation of an acetic acid molecule when a proton is accepted by an. Thermodynamic equilibrium is an axiomatic concept of thermodynamicsIt is an internal state of a single thermodynamic system or a relation between several thermodynamic systems connected by more or less permeable or impermeable wallsIn thermodynamic equilibrium there are no net macroscopic flows of matter or of energy either within a system or between systems.
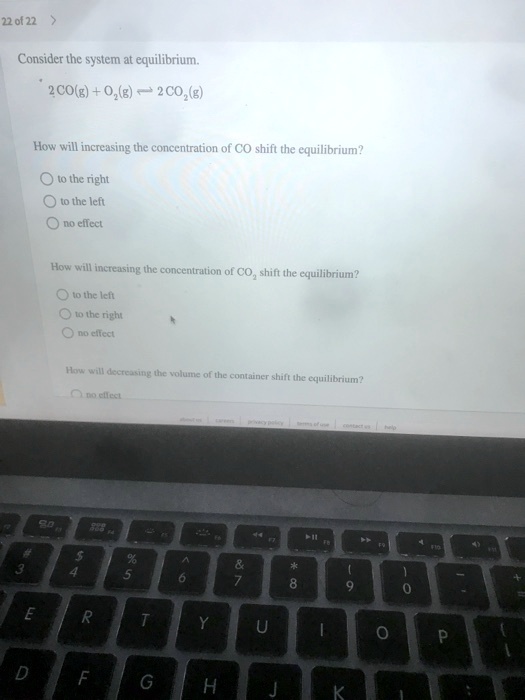
The left junction is shown in Fig. Intuitively this means that if any given player were told the strategies of all their opponents they still would choose to retain their original strategy. Or if we remove reactants from the system equilibrium will also be shifted to the left.
Equilibrium in physics the condition of a system when neither its state of motion nor its internal energy state tends to change with time. If the system moves away from the equilibrium after small disturbances then the equilibrium is unstable.
Consider the two-dimensional autonomous system f1xy -y y-1 y f2xy x-1y-1 a Find all of the equilibrium points.
Since this junction in the strings is in. Economic equilibrium is a condition or state in which economic. Thermodynamic equilibrium is an axiomatic concept of thermodynamicsIt is an internal state of a single thermodynamic system or a relation between several thermodynamic systems connected by more or less permeable or impermeable wallsIn thermodynamic equilibrium there are no net macroscopic flows of matter or of energy either within a system or between systems. Thermodynamics is the study of the relationship between heat temperature work and energy. For example in the game of trying to guess 23 of the average guesses the unique Nash equilibrium is. B Sketch the zero curves fixy 0 fzx y 0. Chapter 11 Rotational Dynamics and Static Equilibrium Q24P Consider the system in Active Example 11-1 this time with the axis of rotation at the location of the child. Intuitively this means that if any given player were told the strategies of all their opponents they still would choose to retain their original strategy. The 50 N force is not equal to the 30 N force.
Most generally equilibrium refers to a balanced state that does not change overall with. Most generally equilibrium refers to a balanced state that does not change overall with. Learn about Kc and Kp here. Chemical equilibrium may also be called a steady state reaction This does not mean the chemical reaction has necessarily stopped occurring but that the consumption and formation of substances have reached a balanced. Vapor-liquid equilibrium VLE data can be computed using the modified Raoults law. Two physical systems are in thermal equilibrium if there is no net flow of thermal energy between them when they are connected by a path permeable to heat. Equilibrium with respect to a single commodity when supply and demand in that market depend on the prices of other goods.






































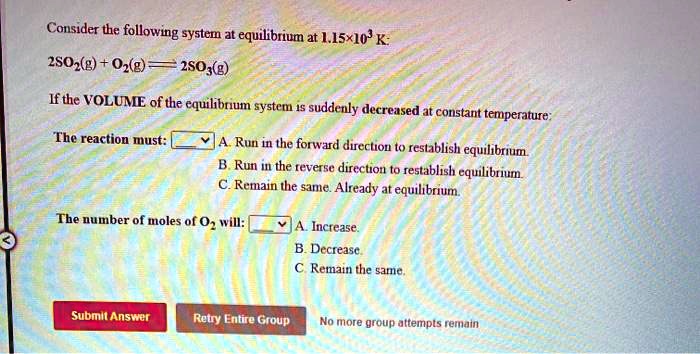

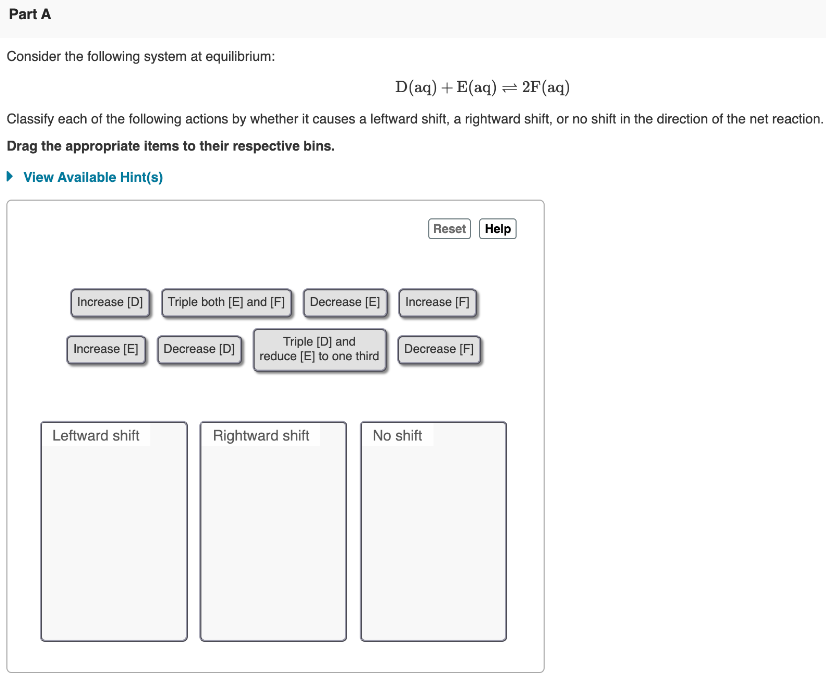
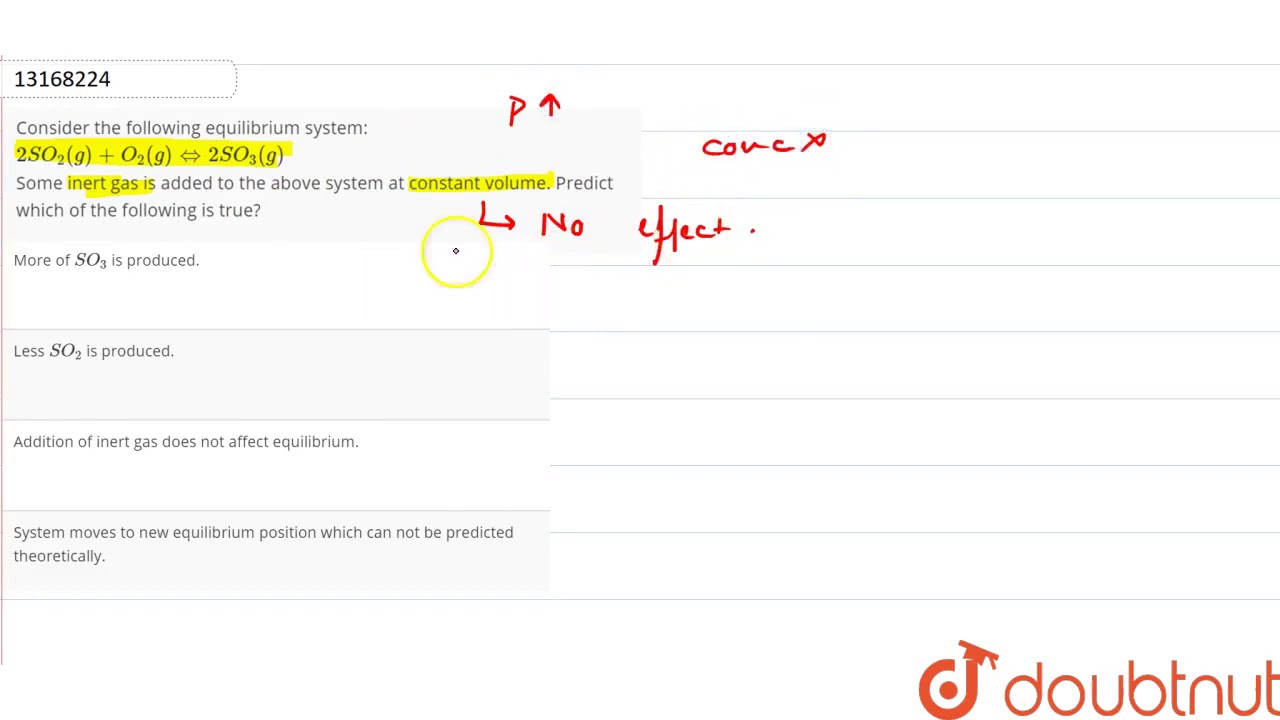
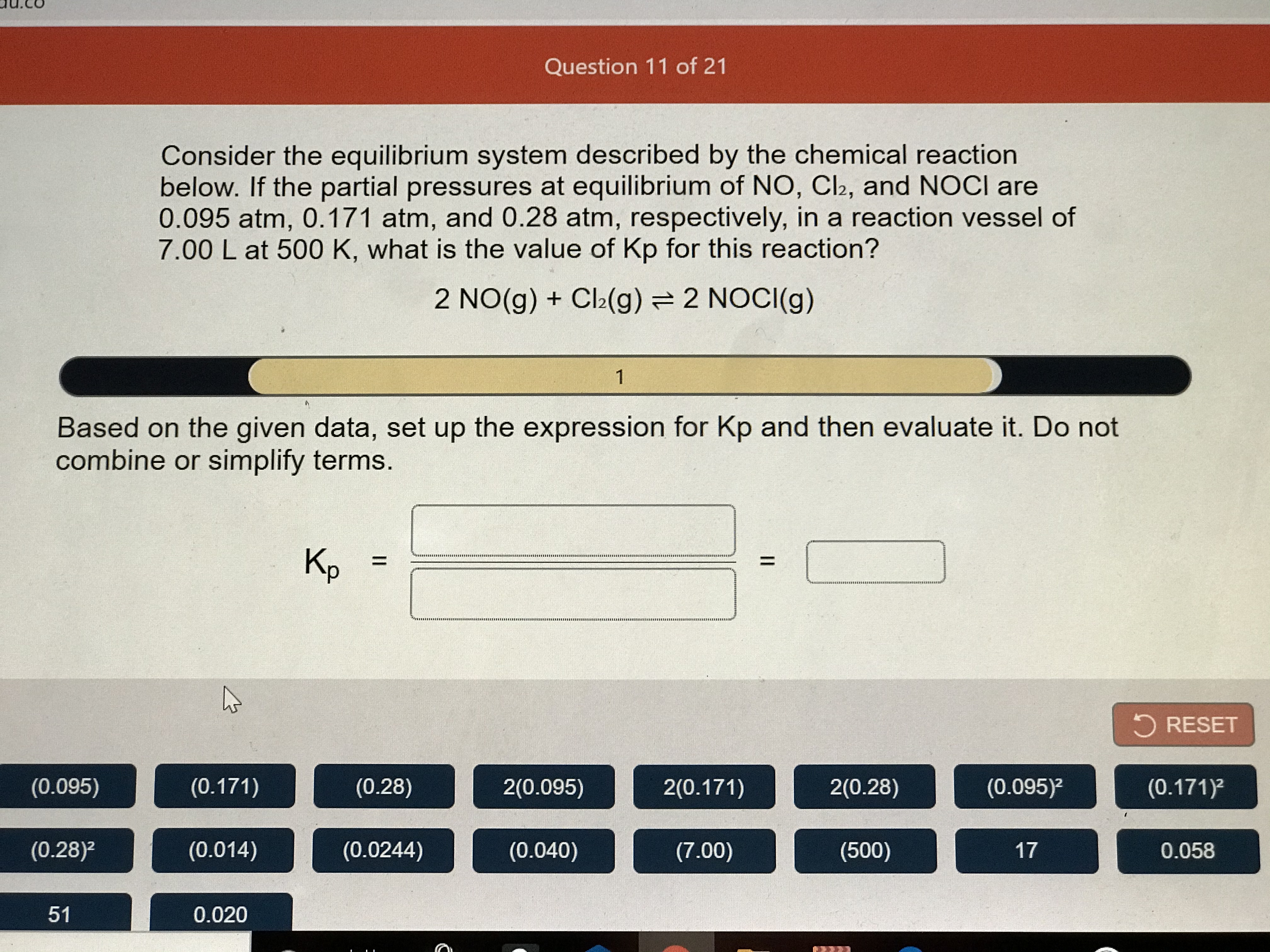

Post a Comment for "Consider This System At Equilibrium"